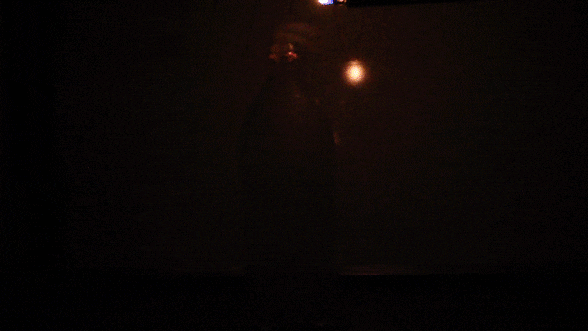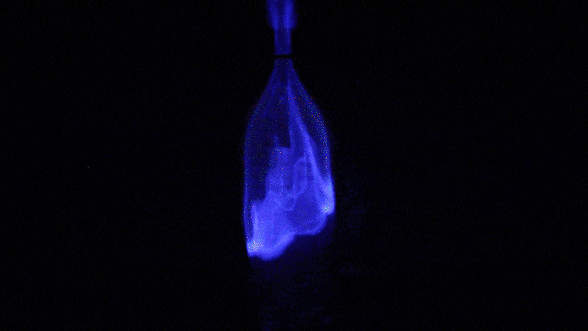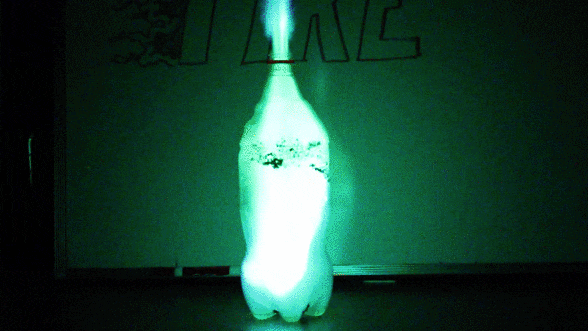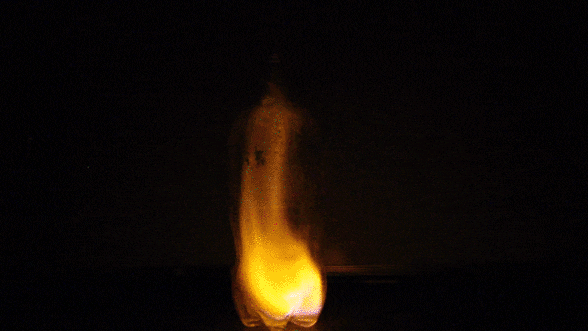This isn’t your typical cloud, and not just because it’s trapped inside a bottle! We chose rubbing alcohol as the raw material for our homemade tabletop cloud because it vaporizes so easily:

Rubbing alcohol is also highly flammable. Let’s explore this property by creating some combustion reactions! Every fire (or combustion reaction) requires fuel, an oxidizing agent (like oxygen), and activation energy. Activation energy is the trigger that causes the combustion reaction to start.
In this experiment, we touch a flame to the top of a cloud of fuel (rubbing alcohol vapor). The vapor is suspended in air, so there’s plenty of oxygen present. Heat from the lighter causes the closest alcohol molecules to react with nearby oxygen molecules. As they react, their atoms recombine to form carbon dioxide and water vapor. These products tie up less energy than the original materials; the leftover energy is released in the form of visible light and heat. Newly generated heat provides activation energy for the next layer of combustion reactions, and the process continues until all of the fuel has been consumed.

Isopropyl alcohol naturally burns blue, but it’s easy to change the hue of a flame. Just add salt! What our eyes interpret as colors are really waves of light stretched out to varying degrees. We say that the most stretched-out waves have the longest wavelength. We call the longest wavelengths we can see “red” and the shortest “violet”. A substance’s chemistry determines the wavelength– and thus the color– of the light that will be released by its combustion. Copper-based salts burn green. Strontium turns flames bright red. Sodium salts, such as table salt, burn yellow. If you’ve ever seen a multicolored fireworks show, you’ve experienced this science firsthand!